EFFECTS OF DIETARY CARBOHYDRATE AND LIPID LEVELS ON GROWTH PERFORMANCE AND PLASMA METABOLITES IN JUVENILE BLUNT SNOUT BREAM
-
摘要: 实验以初重(6.20±0.01) g的团头鲂(Megalobrama amblycephala)幼鱼为研究对象, 分别投喂高糖低脂(HCLL, 45%糖和2%脂)、中糖中脂(MCML, 30%糖和8%脂)和低糖高脂(LCHL, 15%糖和14%脂)的3种等氮饲料56d, 以探究饲料不同糖和脂水平对团头鲂生长性能、鱼体生化组成、营养沉积及血浆代谢物的影响。实验结果显示, 饲料糖和脂水平对团头鲂摄食率无显著性影响。随饲料糖水平降低和脂水平升高, 团头鲂幼鱼特定生长率和饲料效率提高。高糖低脂饲料未造成肝脏糖原或甘油三酯含量的显著沉积, 且各饲料组间肝体比、脏体比及肥满度均无显著性差异。饲料糖和脂水平对团头鲂鱼体生化组成无显著性影响, 但随饲料糖水平降低和脂水平增加, 团头鲂蛋白沉积率升高, 脂肪沉积率降低。另外, 血浆葡萄糖、甘油三酯、游离脂肪酸和胆固醇含量各组间均无显著性差异。低糖高脂组血浆低密度脂蛋白胆固醇显著升高, 表明肝脏将脂质转运至外周组织以维持肝脏脂稳态。研究结果表明, 团头鲂对饲料脂利用优于糖类, 且具有较好地应答饲料糖和脂水平的代谢机制, 研究结果可为团头鲂的饲料配方设计提供支撑。Abstract: To test the effects of dietary carbohydrate and lipid levels on growth performance, body composition, nutrient retention and plasma metabolites in juvenile blunt snout bream (Megalobrama amblycephala) [initial weight of (6.20±0.01) g], fish were fed with three isonitrogenous diets: HCLL (45% carbohydrate, 2% lipid), MCML (30% carbohydrate, 8% lipid), or LCHL (15% carbohydrate, 14% lipid) for 56 days. Results showed that dietary carbohydrate and lipid had no effect on FR, but declined dietary carbohydrate and increased lipid enhanced the SGR and FE. The HCLL diet did not induce the retention of glycogen and TG in the liver, nor did it alter morphological indices like HSI, VSI or CF. In addition, no significant differences were observed on the body composition of blunt snout bream fed with varying dietary carbohydrate and lipid levels, but as the dietary carbohydrate reduced and lipid increased the PRE elevated and LRE declined. Furthermore, no significant differences were found on the plasma glucose, TG, free fatty acids and cholesterol levels among all experiment group. The LCHL diet significantly induced the plasma LDL-C content, suggesting the liver transferred lipid to peripheral tissue to sustain the lipid homeostasis. In conclusion, the blunt snout bream can utilize lipid better than carbohydrate. Meanwhile, the blunt snout bream has an adaptive system that can cope with changes in dietary carbohydrate and lipid level. The present study can provide information for blunt snout bream feed formulation.
-
Keywords:
- Blunt snout bream /
- Carbohydrate /
- Lipid /
- Growth performance /
- Nutrient retention /
- Plasma metabolites
-
饲料蛋白质是维持鱼体组织生长和生命功能的重要物质, 也是价格最高的饲料原料[1]。在所有的蛋白源中, 鱼粉因其氨基酸组成平衡, 可消化性强和含有较少的抗营养因子, 被认为是水产饲料的优质蛋白源[2]。然而鱼粉的生产依赖于海洋鱼类资源, 水产饲料行业的可持续性发展需要进一步降低海洋资源的使用[3], 因此增加饲料中非蛋白能源物质的比例是目前研究的热点。糖类和脂肪作为重要的非蛋白能源物质, 被认为具有节约蛋白的作用, 在水产行业领域中的重要性与日俱增[4, 5]。
糖类来源广泛、价格低廉, 是饲料的主要能量来源[5, 6]。糖类有利于饲料的制粒加工, 减少蛋白质作为能量的消耗, 并且可以为许多生物合成过程提供代谢底物[6, 7]。然而, 饲料糖类含量过高则会导致许多的负面效应, 如生长受阻、脂肪的异常沉积和免疫机能下降等[8—10]。饲料脂肪为鱼类生长发育提供能量, 同时为机体提供必需脂肪酸和一些脂溶性维生素, 对于鱼类生命活动具有重要意义[11, 12]。脂肪可以被大多数鱼类较好的利用, 但饲料脂肪含量过剩或短缺都会对鱼类生长和免疫造成直接的影响[13, 14]。此外, 糖类和脂肪利用相互影响, 密不可分。脂类物质可以通过糖异生途径转化为葡萄糖, 糖也可以为脂肪合成提供底物从而转化为脂肪储存在组织中[15]。因此, 饲料中适宜的糖和脂肪水平对水产养殖的发展具有重要意义。在饲料糖和脂相对水平对鱼类影响的研究中, 饲料糖脂比在不同种类如长吻(Leiocassis longirostris Günther)[16]、黄颡鱼(Pelteobagrus fulvidraco)[17]、大菱鲆(Scophthalmus maximus)[18]、草鱼(Ctenopharyngodon idella)[8]和罗非鱼(Oreochromis niloticus)[19]等鱼类中均有开展。以上研究内容旨在探究不同鱼类生长或生理状态最佳条件下的最适糖脂比, 其确定可为饲料配比提供理论依据。
团头鲂(Megalobrama amblycephala)是我国重要的草食性淡水鱼类, 具有生长快速、肉质鲜美和存活率高等优点[20, 21]。团头鲂在我国消费需求量大, 2019年的年产量已达78.32×107
kg[22]。关于团头鲂糖和脂需求方面的研究较多, 研究结果表明, 团头鲂幼鱼最适饲料糖和脂需求分别为31%—34%和8%[23, 24]。但相关研究较多为单一性的不同含量或不同来源的糖或脂肪的研究[23-27]。有关团头鲂糖脂比的研究均是按照糖/脂的比值按一定的比例逐渐增加, 来探究最适生长或生理状态的糖/脂比值。研究表明高糖饲料促进了肝脏糖酵解过程, 在以特定生长率为评判指标时, 团头鲂最适糖脂比为3.58(糖和脂含量分别为291.70和81.40 g/kg)[28]。在以应激和抗氧化反应为评判指标时, 糖脂比在5.64(糖和脂含量分别为320.87和58.30 g/kg)时血浆皮质醇和乳酸等应激生物标志物含量最低[9]。但是文章中并没有对脂肪代谢及糖和脂肪的交互作用进行研究。本研究以适合的糖和脂含量(中糖中脂)为对照, 探究在高糖低脂和低糖高脂这两种相差较大的糖和脂的配比模式下, 糖和脂交互作用对团头鲂生长性能、饲料利用、营养沉积及相关生理代谢物的影响。 1. 材料与方法
1.1 饲料配方与制作
本实验制备了3种等氮饲料(蛋白质含量32%), 分别包含不同糖和脂水平: 高糖低脂(HCLL, 45%糖和2%脂)、中糖中脂(MCML, 30%糖和8%脂)及低糖高脂(LCHL, 15%糖和14%脂)饲料。实验饲料以玉米淀粉为碳水化合物源, 鱼油和豆油等量混合为脂肪源, 饲料配方如表 1所示。对所有饲料原料都进行研磨, 均匀混合后过40目筛网后加水调制, 用制粒机(SLP-45, 渔业机械设备研究所, 中国上海)制备成饲料颗粒。以上各颗粒饲料于烘箱50℃烘干后于4℃冰柜中保存备用。其中, 饲料的脂肪酸组成见表 2。
表 1 饲料配方和化学组成(% 风干重)Table 1. Formulation and chemical composition of experimental diets原料Ingredient (% dry matter) 饲料Diet 高糖低脂HCLL 中糖中脂MCML 低糖高脂LCHL 白鱼粉White fishmeal1 15 15 15 酪蛋白Casein2 24 24 24 鱼油﹕豆油Fish oil3﹕
Soybean oil (1﹕1)0 6 12 玉米淀粉Corn starch4 45 30 15 维生素预混物Vitamin premix5 0.39 0.39 0.39 氯化胆碱Choline chloride 0.11 0.11 0.11 矿物盐预混物Mineral premix6 5 5 5 羧甲基纤维素钠CMC Carboxymethylcellulose sodium 3 3 3 纤维素Cellulose 7.5 16.5 25.5 化学组成Chemical composition (%) 粗蛋白Crude protein 31.92 31.82 31.72 粗脂肪Crude lipid 2.21 8.21 14.21 能量Gross energy (kJ/g) 18.14 19.50 20.86 注: 1白鱼粉: 白鱼粉, 美国海鲜公司; 2酪蛋白: 兰州陇朊干酪素有限责任公司; 3鱼油: 秘鲁鱼油, 购自武汉高龙饲料有限公司; 4玉米淀粉: 武汉高龙饲料有限公司; 5维生素预混物 (mg/kg 饲料): 维生素A, 1.65; 维生素D, 0.025; 维生素E, 50; 维生素K, 10; 维生素C, 100; 硫胺素, 20; 核黄素, 20; 维生素B6, 20; 维生素B12, 0.02; 叶酸, 5; 泛酸钙, 50; 肌醇, 100; 烟酸, 100; 生物素, 0.1; 纤维素, 645.2; 6矿物盐预混物(mg/kg 饲料): 氯化钠, 500; 七水合硫酸镁, 8155.6; 二水合磷酸二氢钠, 12500.0; 磷酸二氢钾, 16000.0; 二水合磷酸氢钙, 7650.6; 七水合硫酸亚铁, 2286.2; 五水合乳酸钙, 1750.0; 七水合硫酸锌, 178.0; 一水合硫酸锰, 61.4; 五水合硫酸铜, 15.5; 七水合硫酸钴, 0.5; 碘化钾, 1.5; 玉米淀粉, 753.7Note: 1White fishmeal: White fishmeal, American Seafood Company, Seattle, Washington, USA; 2 Casein: Lanzhou Longruan Casein Co. Ltd., Lanzhou, Gansu, China; 3 Fish oil: Anchovy oil from Peru, purchased from Coland Feed Co., Ltd., Wuhan, China; 4 Corn starch: Wuhan Coland Feed Co., Ltd., Wuhan, China; 5 Vitamin premix (mg/kg diet): Vitamin A, 1.65; Vitamin D, 0.025; Vitamin E, 50; Vitamin K, 10; Vitamin C, 100; Thiamin, 20; Riboflavin, 20; Pyridoxine, 20; Cyanocobalamin, 0.02; Folic acid, 5; Calcium patotheniate, 50; Inositol, 100; Niacin, 100; Biotin, 0.1; Cellulose, 645.2; 6Mineral premix (mg/kg diet): NaCl, 500; MgSO4·7H2O, 8155.6; NaH2PO4·2H2O, 12500.0; KH2PO4, 16000.0; CaHPO4·H2O, 7650.6; FeSO4·7H2O, 2286.2; C6H10CaO6·5H2O, 1750.0; ZnSO4·7H2O, 178.0; MnSO4·H2O, 61.4; CuSO4·5H2O, 15.5; CoSO4·7H2O, 0.5; KI, 1.5; Corn starch, 753.7 表 2 饲料脂肪酸组成(% 总脂肪酸)Table 2. Fatty acid composition of the experimental diets (% of total fatty acids)脂肪酸Fatty acid (%) 饲料Diet 高糖低脂
HCLL中糖中脂
MCML低糖高脂
LCHLC14﹕0 3.27 4.71 6.08 C15﹕0 0.36 0.46 0.56 C16﹕0 13.44 11.05 14.41 C17﹕0 0.28 0.35 0.22 C18﹕0 6.40 5.85 7.16 ΣSFA1 23.75 22.43 28.44 C16﹕1n-9 5.71 4.48 6.40 C18﹕1n-9 36.27 26.21 0.13 C20﹕1n-9 3.54 2.63 2.90 C22﹕1n-9 2.71 4.04 5.28 ΣMUFA2 48.23 37.35 14.71 C18﹕2n-6 15.18 24.00 36.76 C18﹕3n-6 0.23 0.14 0.16 C20﹕2n-6 0.43 0.35 0.33 C20﹕4n-6 0.60 0.77 0.03 Σn-6 PUFA3 16.43 25.26 37.29 C18﹕3n-3 0.76 3.37 5.18 C20﹕5n-3 2.69 4.31 6.26 C22﹕6n-3 4.72 5.76 6.75 Σn-3 PUFA 8.17 13.43 18.19 n-3/n-6 PUFA 0.50 0.53 0.49 注: 1SFA: 饱和脂肪酸; 2 MUFA: 单不饱和脂肪酸; 3多不饱和脂肪酸Note: 1SFA: saturated fatty acids; 2MUFA: monounsaturated fatty acids; 3PUFA: polyunsaturated fatty acids 1.2 饲养管理
实验所用团头鲂幼鱼来自中国科学院水生生物研究所(中国武汉)。在正式养殖实验前, 将制备的3种饲料等量均匀混合投喂进行为期2周的驯养以适应实验系统环境。实验开始前对实验鱼饥饿24h后捞出, 随机挑选规格均匀和体质健康的团头鲂幼鱼[初重(6.20±0.01) g]分为3个处理组。其中每缸30尾鱼, 每个处理组3个平行。实验在9个圆柱形玻璃纤维缸(水体积为167 L)组成的室内循环水系统中进行, 生长实验持续56d, 每天饱食投喂3次(08:30、13:30和18:30)并记录摄食量和残饵。实验期间每天进行水质监测管理, 其中水温为25—27℃, 溶氧(7.09±0.12) mg/L, 氨氮(0.18±0.02) mg/L, pH为6.5—7.0。光照强度为2.21—2.95 μmol/(s·m2)。光照周期为12L﹕12D(8:00—20:00光亮)。
1.3 样品采集
在养殖实验结束后, 在最后一次投喂后8h进行取样。实验鱼用麻醉剂MS-222 (60 mg/L, Sigma, 美国) 麻醉后称总重后取样。每缸4尾鱼称重后保存于–20℃冰箱中用于体成分分析。每缸3尾鱼进行体长和体重测量, 并解剖肝脏和内脏团称重以计算肝体比和脏体比。另外, 每缸随机取2尾鱼, 用肝素钠抗凝剂润过的无菌注射器进行尾部静脉采血。血液3000×g 10min离心后得到血浆, 保存于–80℃。同时, 在冰上取出肝脏组织, 置于液氮中速冻并于–80℃保存。
1.4 样品分析
本实验所有饲料和鱼样样品基本组成的测定均参照AOAC (2003) 标准方法[29]进行检测。干物质通过在烘箱(电热恒温干燥箱, 精宏, 中国上海)中105℃烘干至恒重, 通过失重法测定; 灰分在马弗炉(马弗炉, 中国湖北)中550℃煅烧3h, 采用失重法测定; 粗蛋白采用凯氏定氮仪(2300, Kjeltec Analyzer Unit)测定; 粗脂肪采用索氏抽提仪(Soxtec System HT6, Tecator, Hoganas, Sweden)进行抽提测定; 饲料脂肪酸含量参照Folch等[30]方法采取氯仿甲醇 = 2﹕1混合试剂提取总脂, 进行脂肪酸甲酯化处理后采用气相色谱仪测定脂肪酸组成和含量 (7890A-5975C, Agilents Technologies Inc., Santa Clara, CA, USA)。
肝糖原含量根据南京建成生物工程研究所糖原测试盒(A043-1-1)说明书进行, 该方法根据蒽酮法原理, 利用糖原在浓硫酸作用下可脱水生成醛糖衍生物, 后者再与蒽酮作用形成蓝色复合物, 与同法处理的标准葡萄糖溶液比色定量测得。肝脏甘油三酯(TG)含量采用GPO-PAP方法, 由南京建成采用商品试剂盒(A100-1-1)检测(南京建成生物工程研究所, 中国南京)。
血浆葡萄糖、甘油三酯、游离脂肪酸(NEFA)和胆固醇含量采用商品试剂盒进行测定(Fujifilm, Wako Pure Chemical, Corporation, Osaka, Japan)。血浆低密度脂蛋白胆固醇(LDL-C)和高密度脂蛋白胆固醇(HDL-C)分别采用南京建成商品试剂盒A113-1-1和A112-1-1检测(南京建成生物工程研究所, 中国南京)。
1.5 数据统计分析
本实验所有数据使用统计软件SPSS 18.0进行统计分析。实验结果采用均值±标准误(Mean±SEM, n=6), 进行单因素方差分析(One-way ANOVA)。若各实验组间差异显著, 则同质性检验后进行Duncan’s多重比较。以P<0.05表示统计学差异显著。
2. 结果
2.1 生长与饲料利用
如表 3所示, 摄食不同糖和脂水平饲料, 团头鲂幼鱼摄食率FR各组间无显著性差异(P>0.05), 但鱼均摄食量FI在HCLL组显著低于MCML组(P<0.05)。团头鲂摄食HCLL饲料组特定生长率SGR显著低于MCML和LCHL组(P<0.05)。团头鲂饲料效率随饲料糖水平减少脂水平增多而升高, 在高糖低脂HCLL组最低。摄食不同糖和脂水平饲料对团头鲂肥满度CF、肝体比HSI、脏体比VSI均无显著性差异(P>0.05)。
表 3 不同饲料糖和脂水平对团头鲂幼鱼生长性能、饲料利用和形体指数的影响Table 3. Effects of dietary carbohydrate and lipid levels on growth performance, feed utilization, and morphological indices in blunt snout bream juveniles指标Index 饲料Diet 高糖低脂HCLL 中糖中脂MCML 低糖高脂LCHL 初始体重IBW (g) 6.02±0.01 6.02±0.02 6.02±0.01 终末体重FBW (g) 23.18±1.85a 33.48±1.89b 31.93±2.96b 摄食率FR1 (% BW/d) 2.93±0.26 3.04±0.13 2.76±0.04 摄食量FI2 (g/fish) 23.96±2.62a 32.28±0.46b 28.81±1.77ab 饲料效率FE3 (%) 71.98±3.74a 81.54±1.63ab 87.69±2.67b 特定生长率SGR4 (%/d) 2.40±0.14a 3.06±0.10b 2.96±0.17b 肥满度CF5 (g/cm3) 2.27±0.03 2.45±0.07 2.52±0.09 肝体比HSI6 2.21±0.23 1.77±0.14 1.30±0.24 脏体比VSI7 10.59±0.54 10.50±0.31 10.79±0.40 注: 表中所示数据表示为平均值±标准误, 同行数值上标英文字母不同则表示差异性显著 (P<0.05); 下同; 1摄食率FR (% BW/d)=100×干物质摄食量/[天数×(初始体重+终末体重)/2]; 2摄食量FI (g/fish)=总干物质摄食量/鱼数目; 3饲料效率 FE (%)= 100×鱼体总增重/摄食量; 4特定生长率 SGR (%/d)=100×[ln (末重)–ln (初重)]/天数; 5肥满度CF (g/cm3)=100×体重/体长3; 6肝体比HSI=100×肝脏重/体重; 7脏体比VSI=100×内脏重/体重Note: Values are presented as Mean±SEM. The same applies below. Values in the same row with different superscript letters are significantly different (P<0.05). 1Feeding rate (FR, % BW/d)=100×dry feed intake/[days×(IBW+FBW)/2]; 2Feed intake (FI, g/fish)=dry feed intake/number of fish; 3Feed efficiency (FE, %)=100×fresh body weight gain/dry feed intake; 4Specific growth rate (SGR, %/d)=100×[ln (final weight)–ln (initial weight)]/days; 5Condition factor (CF, g/cm3)=100×(body weight)/(body length)3; 6Hepatosomatic index (HSI)=100×liver weight/whole body weight; 7Viscerosomatic index (VSI)=100×viscera weight/whole body weight 2.2 鱼体组成与营养沉积
如表 4所示, 团头鲂幼鱼摄食不同糖和脂水平饲料后, 其全鱼粗蛋白、粗脂肪、水分及灰分含量均无显著性差异(P>0.05)。随饲料糖含量增加脂含量减少, 鱼体蛋白沉积率降低, 反之, 脂肪沉积率增加。
表 4 不同饲料糖和脂水平对团头鲂幼鱼体生化组成和营养沉积的影响Table 4. Effects of dietary carbohydrate and lipid levels on body chemical composition and nutrient retention efficiency in blunt snout bream juveniles指标Index 饲料Diet 高糖低脂HCLL 中糖中脂MCML 低糖高脂LCHL 粗蛋白Crude protein (%) 14.79±0.75 15.82±0.22 15.51±0.03 粗脂肪Crude lipid (%) 8.36±0.66 10.43±0.38 10.12±0.44 灰分Ash (%) 2.98±0.14 3.11±0.03 2.97±0.06 水分Moisture (%) 73.51±1.51 70.22±0.43 70.82±0.51 蛋白沉积率PRE1 (%) 36.56±1.85a 40.22±0.47a 45.70±0.91b 脂肪沉积率LRE2 (%) 354.93±8.36c 114.81±6.87b 71.58±4.65a 注: 1蛋白质沉积率PRE(%)=100×鱼体蛋白质沉积量/蛋白质摄入量; 2脂肪沉积率LRE(%)=100×鱼体脂肪沉积量/脂肪摄入量Note: 1Protein retention efficiency (PRE, %)=100×body retained protein/protein intake; 2Lipid retention efficiency (LRE, %)=100×body retained lipid/lipid intake 2.3 肝脏糖原和甘油三酯含量
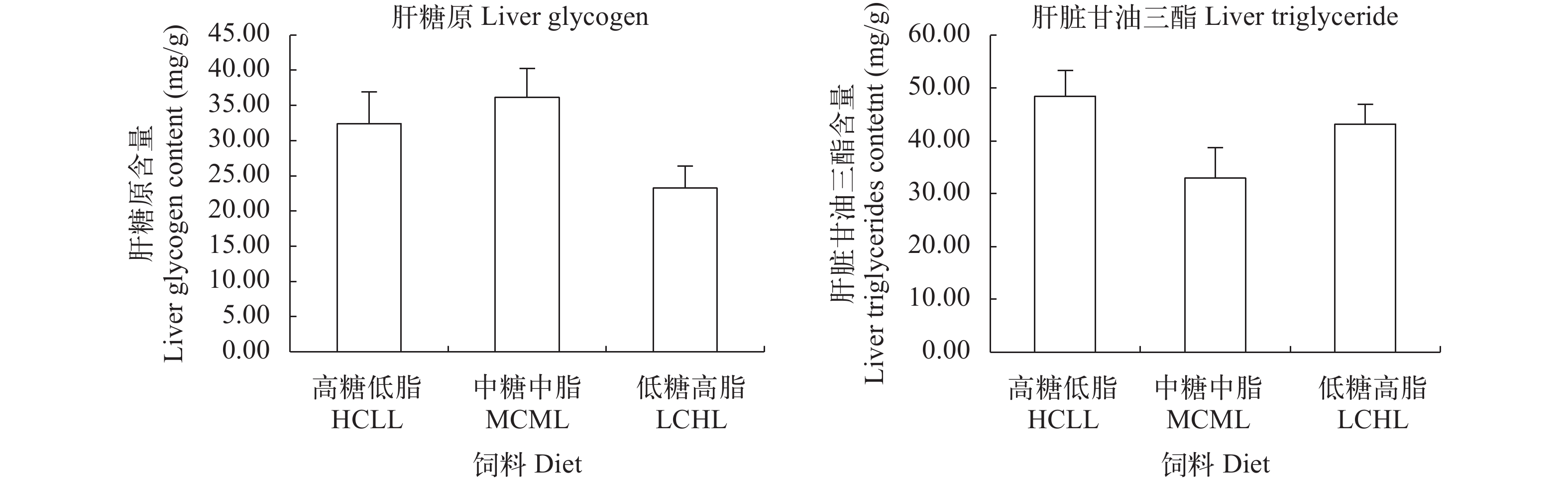
如图 1所示, 不同糖和脂水平饲料组间, 团头鲂肝糖原和甘油三酯含量均无显著性差异(P>0.05)。
2.4 血浆代谢物水平
如表 5所示, 血浆葡萄糖水平、甘油三酯、游离脂肪酸、总胆固醇及高密度脂蛋白胆固醇含量在各饲料组间均无显著性差异(P>0.05)。血浆低密度脂蛋白胆固醇含量在LCHL组显著低于其他饲料组(P<0.05)。
表 5 不同饲料糖和脂水平对团头鲂幼血浆生化组成的影响Table 5. Effects of dietary carbohydrate and lipid levels on plasma metabolites in blunt snout bream juveniles指标Index 饲料Diet 高糖低脂HCLL 中糖中脂MCML 低糖高脂LCHL 血糖Glucose (mmol/L) 3.35±0.56 3.43±0.37 4.10±0.37 甘油三酯Triglycerides (mmol/L) 2.50±0.35 2.62±0.24 3.45±0.32 游离脂肪酸Free fatty acids (mEq/L) 0.67±0.11 0.81±0.14 0.51±0.06 胆固醇Cholesterol (mmol/L) 7.78±0.64 8.96±0.60 6.87±0.29 低密度脂蛋白LDL-C (mmol/L) 8.06±0.33b 8.26±0.45b 5.78±0.62a 高密度脂蛋白HDL-C (mmol/L) 6.47±0.18 7.19±0.31 7.33±0.52 3. 讨论
3.1 饲料糖和脂水平对团头鲂生长和饲料利用的影响
饲料中非蛋白能源物质糖和脂肪的适宜添加水平对于维持鱼类生长和生命过程十分重要, 但过高或过低的添加水平都会对鱼体造成负面的生理响应[6, 8, 31]。高糖饲料导致的生长抑制效应在多种鱼类中已经得到证实, 如虹鳟(Salmo gairdneri)[32]、大西洋鲑(Salmo salar L.)[33]和草鱼(Ctenopharngon idellus)[34]等。在团头鲂糖脂比的研究中, 团头鲂摄食量随饲料糖脂比升高(CHO/L从1.62到10.46)而增多, 但更高的糖脂比(CHO/L: 24.20)却导致摄食量的显著下降, 从而抑制了团头鲂的生长[28]。与此一致, 在本实验中团头鲂高糖低脂HCLL组(CHO/L: 22.5)鱼均摄食量FI显著低于适宜糖脂中糖中脂MCML组, 且高糖低脂HCLL饲料显著降低了团头鲂幼鱼的饲料利用效率, 造成特定生长率下降。由此可见, 高糖造成了团头鲂生长性能受到抑制的负面效应。另外, 研究表明饲料高小麦淀粉水平造成草鱼对蛋白和糖的表观消化率显著低于低淀粉水平组[34], 表明高糖可能造成鱼类对营养物质消吸收的减弱, 也可能是造成生长抑制的原因之一。然而, 鱼类对饲料中糖和脂肪的利用受到两者水平的相互影响, 饲料的脂肪水平也会影响糖的利用[15, 35], 前期已有研究验证了团头鲂对于糖和脂的利用存在交互影响[36]。并且, 饲料脂肪含量过低可能不足以为鱼体提供生长必需的能量和某些生物合成过程所需的脂肪酸, 进而导致鱼体的代谢紊乱。在本实验中, 3种糖脂水平饲料的脂肪源均采用鱼油和豆油等比例混合, 脂肪酸种类均衡(表 2), 满足团头鲂对于脂肪酸种类的需求。且已有研究报道团头鲂的最适脂肪需求为8%[23, 24], 在本实验中2%脂肪水平的高糖低脂HCLL组团头鲂摄食量FI和相应的总脂肪摄入量均低于适宜糖脂即中糖中脂MCML组, 不足以维持团头鲂生长所必需的脂肪需求。综上, 高糖和低脂的交互作用因素导致团头鲂摄食量FI和饲料效率FE的降低, 从而抑制了团头鲂的生长性能。另外, 不同鱼类利用糖和脂供能的效率可能不同, 如虹鳟[37]和欧洲海鲈(Dicentrarchus labrax L.)[38]对饲料脂肪的利用优于糖类, 而尼罗罗非鱼(Oreochromis niloticus)[39]和非洲鲶(Clarias gariepinus)[40] 则相反。在本实验3个饲料组中, 随着饲料糖水平降低、脂水平升高, 团头鲂的饲料效率呈升高趋势。这表明团头鲂对脂肪有着更好的利用, 从某种程度上来说其对高糖缺乏耐受[24, 28]。另外, 中糖中脂组特定生长率最高, 表明适宜水平的糖和脂的配伍才可以达到团头鲂生长性能的最大化, 同时也验证了鱼类饲料糖和脂等营养物质最适需求研究的必要性。
3.2 高糖低脂未造成团头鲂肝脏脂肪异常沉积或形体指数的改变
一般来说, 摄入过量的碳水化合物, 其消化产生的单糖会通过糖酵解途径转化为丙酮酸, 进而通过氧化为机体供能或者合成脂肪在机体中储存起来[41]。因此, 高糖饲料通常造成许多鱼类肝体比增加, 肝脏脂肪代谢的紊乱甚至是脂肪肝的生成[8, 42]。在本实验中, 3种糖和脂水平饲料组间团头鲂幼鱼肝糖原含量和甘油三酯含量无显著性差异, 且肝体比各组间也无显著性差异。因此, 高糖低脂饲料并未造成团头鲂肝脏脂肪或糖原的异常沉积或脂肪肝的产生。与本实验结果一致, 在团头鲂不同糖脂比的研究中, 饲料糖脂比24.2时即糖41.6%、脂1.7%含量时肝体比HSI和肝脏脂肪含量与其他组间无显著性差异[28]。这一结果的产生可能归因于以下几点: 首先, 肝脏不是团头鲂脂肪沉积的主要场所[28, 36]; 其次, 在刘梅珍等[43]对团头鲂的研究中, 2%—11%的不同脂肪水平饲料都未诱发团头鲂的肝脏脂肪沉积, 所以本实验高糖低脂HCLL饲料中2%的脂肪含量对于诱发团头鲂的脂肪沉积较低。综合以上因素, 高糖低脂HCLL饲料对团头鲂形体指数、肝糖原和脂肪沉积无显著性影响。
3.3 饲料糖和脂水平与团头鲂鱼体营养沉积
在一些鱼类中, 饲料糖水平可能比脂水平有着更重要的脂肪合成诱因效果, 高糖饲料可以促进脂肪合成相关酶活性[8, 42]。并且, 在某些鱼类中鱼体脂肪含量随饲料碳水化合物水平升高而升高[38]。在本实验中, 各饲料组团头鲂体粗脂肪含量无显著性差异。但高糖低脂HCLL组脂肪沉积率LRE显著高于其他两饲料组, 表明机体内存在由糖向脂的转化过程。而表观上来讲, 较高的脂肪沉积率却未造成鱼体脂肪生化组成高于其他两饲料组, 这一现象产生的原因可能是高糖低脂HCLL组食源性脂肪摄入量少于其他两饲料组, 高糖转化为的脂肪用于机体生长而未在鱼体内过度沉积。同时, 脂肪作为重要的能源物质, 被认为具有节约蛋白的作用[4]。随着饲料脂肪含量升高, 团头鲂更多的利用脂肪来为机体供能, 从而减少了蛋白作为能量物质用于生长活动的消耗, 促进了低糖高脂组的蛋白沉积。与之一致的是, 团头鲂氮沉积率随饲料糖脂比增加(CHO/L: 2.45—24.0)而减少[28]。由此可知, 尽管团头鲂作为草食性鱼类, 但随饲料脂肪增多可以更好地储存蛋白质, 表明团头鲂对脂肪的利用较好。
3.4 团头鲂脂质转运与脂质稳态调节
肝脏是鱼类中间代谢的主要器官, 且肝脏脂稳态是一个复杂的过程, 包括脂的分泌、转运和摄取等过程的综合作用[13]。血浆生化指标可以作为机体代谢和生理状态的反应, 如甘油三酯、胆固醇和脂蛋白等脂类物质水平可以反映机体对于脂质的动员情况[44, 45]。在本实验中, 团头鲂血浆葡萄糖、甘油三酯、游离脂肪酸和胆固醇的含量各饲料组间无显著性差异, 表明团头鲂在不同糖和脂水平饲料摄入下可以维持血浆生理的稳态。与哺乳动物类似, 鱼类的脂类转运通过脂蛋白经内源性和外源性循环来调控。其中内源性途径是指肝脏中极低密度脂蛋白(VLDL)向外分泌脂类并转化为中间密度脂蛋白(IDL)和低密度脂蛋白, 而高密度脂蛋白负责脂质的逆向转运[46]。摄食高脂饲料造成草鱼[47]、大黄鱼 (Larimichthys crocea)[46]和红鳍东方鲀 (Takifugu rubripes)[48]等鱼血浆低密度脂蛋白的显著增高。在本实验中低密度脂蛋白胆固醇在低糖高脂组显著增加, 表明团头鲂通过低密度脂蛋白将较多摄入的脂质转移到外周组织从而维持肝脏脂质的稳态。
综上所述, 团头鲂在饲料低糖高脂配比时生长性能、饲料利用和蛋白沉积均显著优于低糖高脂条件, 表明虽然是草食性鱼类, 但团头鲂对脂肪的利用仍优于糖类。且在饲料糖和脂的相对配比下, 团头鲂血浆生理稳态, 未造成肝脏脂肪的异常沉积, 肝体指数的增加或体脂肪的异常改变, 表明团头鲂具有较好的应对饲料糖和脂水平变化的自身代谢调控机制。
-
表 1 饲料配方和化学组成(% 风干重)
Table 1 Formulation and chemical composition of experimental diets
原料Ingredient (% dry matter) 饲料Diet 高糖低脂HCLL 中糖中脂MCML 低糖高脂LCHL 白鱼粉White fishmeal1 15 15 15 酪蛋白Casein2 24 24 24 鱼油﹕豆油Fish oil3﹕
Soybean oil (1﹕1)0 6 12 玉米淀粉Corn starch4 45 30 15 维生素预混物Vitamin premix5 0.39 0.39 0.39 氯化胆碱Choline chloride 0.11 0.11 0.11 矿物盐预混物Mineral premix6 5 5 5 羧甲基纤维素钠CMC Carboxymethylcellulose sodium 3 3 3 纤维素Cellulose 7.5 16.5 25.5 化学组成Chemical composition (%) 粗蛋白Crude protein 31.92 31.82 31.72 粗脂肪Crude lipid 2.21 8.21 14.21 能量Gross energy (kJ/g) 18.14 19.50 20.86 注: 1白鱼粉: 白鱼粉, 美国海鲜公司; 2酪蛋白: 兰州陇朊干酪素有限责任公司; 3鱼油: 秘鲁鱼油, 购自武汉高龙饲料有限公司; 4玉米淀粉: 武汉高龙饲料有限公司; 5维生素预混物 (mg/kg 饲料): 维生素A, 1.65; 维生素D, 0.025; 维生素E, 50; 维生素K, 10; 维生素C, 100; 硫胺素, 20; 核黄素, 20; 维生素B6, 20; 维生素B12, 0.02; 叶酸, 5; 泛酸钙, 50; 肌醇, 100; 烟酸, 100; 生物素, 0.1; 纤维素, 645.2; 6矿物盐预混物(mg/kg 饲料): 氯化钠, 500; 七水合硫酸镁, 8155.6; 二水合磷酸二氢钠, 12500.0; 磷酸二氢钾, 16000.0; 二水合磷酸氢钙, 7650.6; 七水合硫酸亚铁, 2286.2; 五水合乳酸钙, 1750.0; 七水合硫酸锌, 178.0; 一水合硫酸锰, 61.4; 五水合硫酸铜, 15.5; 七水合硫酸钴, 0.5; 碘化钾, 1.5; 玉米淀粉, 753.7Note: 1White fishmeal: White fishmeal, American Seafood Company, Seattle, Washington, USA; 2 Casein: Lanzhou Longruan Casein Co. Ltd., Lanzhou, Gansu, China; 3 Fish oil: Anchovy oil from Peru, purchased from Coland Feed Co., Ltd., Wuhan, China; 4 Corn starch: Wuhan Coland Feed Co., Ltd., Wuhan, China; 5 Vitamin premix (mg/kg diet): Vitamin A, 1.65; Vitamin D, 0.025; Vitamin E, 50; Vitamin K, 10; Vitamin C, 100; Thiamin, 20; Riboflavin, 20; Pyridoxine, 20; Cyanocobalamin, 0.02; Folic acid, 5; Calcium patotheniate, 50; Inositol, 100; Niacin, 100; Biotin, 0.1; Cellulose, 645.2; 6Mineral premix (mg/kg diet): NaCl, 500; MgSO4·7H2O, 8155.6; NaH2PO4·2H2O, 12500.0; KH2PO4, 16000.0; CaHPO4·H2O, 7650.6; FeSO4·7H2O, 2286.2; C6H10CaO6·5H2O, 1750.0; ZnSO4·7H2O, 178.0; MnSO4·H2O, 61.4; CuSO4·5H2O, 15.5; CoSO4·7H2O, 0.5; KI, 1.5; Corn starch, 753.7 表 2 饲料脂肪酸组成(% 总脂肪酸)
Table 2 Fatty acid composition of the experimental diets (% of total fatty acids)
脂肪酸Fatty acid (%) 饲料Diet 高糖低脂
HCLL中糖中脂
MCML低糖高脂
LCHLC14﹕0 3.27 4.71 6.08 C15﹕0 0.36 0.46 0.56 C16﹕0 13.44 11.05 14.41 C17﹕0 0.28 0.35 0.22 C18﹕0 6.40 5.85 7.16 ΣSFA1 23.75 22.43 28.44 C16﹕1n-9 5.71 4.48 6.40 C18﹕1n-9 36.27 26.21 0.13 C20﹕1n-9 3.54 2.63 2.90 C22﹕1n-9 2.71 4.04 5.28 ΣMUFA2 48.23 37.35 14.71 C18﹕2n-6 15.18 24.00 36.76 C18﹕3n-6 0.23 0.14 0.16 C20﹕2n-6 0.43 0.35 0.33 C20﹕4n-6 0.60 0.77 0.03 Σn-6 PUFA3 16.43 25.26 37.29 C18﹕3n-3 0.76 3.37 5.18 C20﹕5n-3 2.69 4.31 6.26 C22﹕6n-3 4.72 5.76 6.75 Σn-3 PUFA 8.17 13.43 18.19 n-3/n-6 PUFA 0.50 0.53 0.49 注: 1SFA: 饱和脂肪酸; 2 MUFA: 单不饱和脂肪酸; 3多不饱和脂肪酸Note: 1SFA: saturated fatty acids; 2MUFA: monounsaturated fatty acids; 3PUFA: polyunsaturated fatty acids 表 3 不同饲料糖和脂水平对团头鲂幼鱼生长性能、饲料利用和形体指数的影响
Table 3 Effects of dietary carbohydrate and lipid levels on growth performance, feed utilization, and morphological indices in blunt snout bream juveniles
指标Index 饲料Diet 高糖低脂HCLL 中糖中脂MCML 低糖高脂LCHL 初始体重IBW (g) 6.02±0.01 6.02±0.02 6.02±0.01 终末体重FBW (g) 23.18±1.85a 33.48±1.89b 31.93±2.96b 摄食率FR1 (% BW/d) 2.93±0.26 3.04±0.13 2.76±0.04 摄食量FI2 (g/fish) 23.96±2.62a 32.28±0.46b 28.81±1.77ab 饲料效率FE3 (%) 71.98±3.74a 81.54±1.63ab 87.69±2.67b 特定生长率SGR4 (%/d) 2.40±0.14a 3.06±0.10b 2.96±0.17b 肥满度CF5 (g/cm3) 2.27±0.03 2.45±0.07 2.52±0.09 肝体比HSI6 2.21±0.23 1.77±0.14 1.30±0.24 脏体比VSI7 10.59±0.54 10.50±0.31 10.79±0.40 注: 表中所示数据表示为平均值±标准误, 同行数值上标英文字母不同则表示差异性显著 (P<0.05); 下同; 1摄食率FR (% BW/d)=100×干物质摄食量/[天数×(初始体重+终末体重)/2]; 2摄食量FI (g/fish)=总干物质摄食量/鱼数目; 3饲料效率 FE (%)= 100×鱼体总增重/摄食量; 4特定生长率 SGR (%/d)=100×[ln (末重)–ln (初重)]/天数; 5肥满度CF (g/cm3)=100×体重/体长3; 6肝体比HSI=100×肝脏重/体重; 7脏体比VSI=100×内脏重/体重Note: Values are presented as Mean±SEM. The same applies below. Values in the same row with different superscript letters are significantly different (P<0.05). 1Feeding rate (FR, % BW/d)=100×dry feed intake/[days×(IBW+FBW)/2]; 2Feed intake (FI, g/fish)=dry feed intake/number of fish; 3Feed efficiency (FE, %)=100×fresh body weight gain/dry feed intake; 4Specific growth rate (SGR, %/d)=100×[ln (final weight)–ln (initial weight)]/days; 5Condition factor (CF, g/cm3)=100×(body weight)/(body length)3; 6Hepatosomatic index (HSI)=100×liver weight/whole body weight; 7Viscerosomatic index (VSI)=100×viscera weight/whole body weight 表 4 不同饲料糖和脂水平对团头鲂幼鱼体生化组成和营养沉积的影响
Table 4 Effects of dietary carbohydrate and lipid levels on body chemical composition and nutrient retention efficiency in blunt snout bream juveniles
指标Index 饲料Diet 高糖低脂HCLL 中糖中脂MCML 低糖高脂LCHL 粗蛋白Crude protein (%) 14.79±0.75 15.82±0.22 15.51±0.03 粗脂肪Crude lipid (%) 8.36±0.66 10.43±0.38 10.12±0.44 灰分Ash (%) 2.98±0.14 3.11±0.03 2.97±0.06 水分Moisture (%) 73.51±1.51 70.22±0.43 70.82±0.51 蛋白沉积率PRE1 (%) 36.56±1.85a 40.22±0.47a 45.70±0.91b 脂肪沉积率LRE2 (%) 354.93±8.36c 114.81±6.87b 71.58±4.65a 注: 1蛋白质沉积率PRE(%)=100×鱼体蛋白质沉积量/蛋白质摄入量; 2脂肪沉积率LRE(%)=100×鱼体脂肪沉积量/脂肪摄入量Note: 1Protein retention efficiency (PRE, %)=100×body retained protein/protein intake; 2Lipid retention efficiency (LRE, %)=100×body retained lipid/lipid intake 表 5 不同饲料糖和脂水平对团头鲂幼血浆生化组成的影响
Table 5 Effects of dietary carbohydrate and lipid levels on plasma metabolites in blunt snout bream juveniles
指标Index 饲料Diet 高糖低脂HCLL 中糖中脂MCML 低糖高脂LCHL 血糖Glucose (mmol/L) 3.35±0.56 3.43±0.37 4.10±0.37 甘油三酯Triglycerides (mmol/L) 2.50±0.35 2.62±0.24 3.45±0.32 游离脂肪酸Free fatty acids (mEq/L) 0.67±0.11 0.81±0.14 0.51±0.06 胆固醇Cholesterol (mmol/L) 7.78±0.64 8.96±0.60 6.87±0.29 低密度脂蛋白LDL-C (mmol/L) 8.06±0.33b 8.26±0.45b 5.78±0.62a 高密度脂蛋白HDL-C (mmol/L) 6.47±0.18 7.19±0.31 7.33±0.52 -
[1] Han D, Shan X J, Zhang W B, et al. A revisit to fishmeal usage and associated consequences in Chinese aquaculture [J]. Reviews in Aquaculture, 2018, 10(2): 493-507. doi: 10.1111/raq.12183
[2] Riche M. Nitrogen utilization from diets with refined and blended poultry by-products as partial fish meal replacements in diets for low-salinity cultured Florida pompano, Trachinotus carolinus [J]. Aquaculture, 2015, 435: 458-466. doi: 10.1016/j.aquaculture.2014.10.001
[3] Callet T, Hu H H, Laurence L, et al. Exploring the impact of a low-protein high-carbohydrate diet in mature broodstock of a glucose-intolerant teleost, the rainbow trout [J]. Frontiers in Physiology, 2020, 11: 303. doi: 10.3389/fphys.2020.00303
[4] Watanabe T. Strategies for further development of aquatic feeds [J]. Fisheries Science, 2002, 68(2): 242-252. doi: 10.1046/j.1444-2906.2002.00418.x
[5] Stone D A J. Dietary carbohydrate utilization by fish [J]. Reviews in Fisheries Science, 2003, 11(4): 337-369. doi: 10.1080/10641260390260884
[6] Wilson R P. Utilization of dietary carbohydrate by fish [J]. Aquaculture,, 1994, 124(1–4): 67-80.
[7] Arnesen P, Krogdahl Å. Crude and pre-extruded products of wheat as nutrient sources in extruded diets for Atlantic salmon (Salmo salar, L) grown in sea water [J]. Aquaculture, 1993, 118(1): 105-117.
[8] Gao W, Liu Y J, Tian L X, et al. Effect of dietary carbohydrate-to-lipid ratios on growth performance, body composition, nutrient utilization and hepatic enzymes activities of herbivorous grass carp (Ctenopharyngodon idella) [J]. Aquaculture Nutrition, 2010, 16(3): 327-333.
[9] Li X F, Liu W B, Lu K L, et al. Dietary carbohydrate/lipid ratios affect stress, oxidative status and non-specific immune responses of fingerling blunt snout bream, Megalobrama amblycephala [J]. Fish & Shellfish Immunology, 2012, 33(2): 316-323.
[10] Polakof S, Panserat S, Soengas J L, et al. Glucose metabolism in fish: a review [J]. Journal of Comparative Physiology B, 2012, 182(8): 1015-1045. doi: 10.1007/s00360-012-0658-7
[11] Lin Y H, Shiau S Y. Dietary lipid requirement of grouper, Epinephelus malabaricus, and effects on immune responses [J]. Aquaculture, 2003, 225(1): 243-250.
[12] Li X F, Jiang Y Y, Liu W B, et al. Protein-sparing effect of dietary lipid in practical diets for blunt snout bream (Megalobrama amblycephala) fingerlings: effects on digestive and metabolic responses [J]. Fish Physiology & Biochemistry, 2012, 38(2): 529-541.
[13] Lu K L, Xu W N, Li X F, et al. Hepatic triacylglycerol secretion, lipid transport and tissue lipid uptake in blunt snout bream (Megalobrama amblycephala) fed high-fat diet [J]. Aquaculture, 2013, 408-409(9): 160-168.
[14] Sáez-Royuela M, Casado M, Celada J D, et al. Effect of dietary lipid level on survival, growth performance and body composition of juvenile tench (Tinca tinca L.) fed practical diets [J]. Aquaculture, 2015(439): 14-19. doi: 10.1016/j.aquaculture.2015.01.017
[15] Honorato C A, Almeida L C, Dasilva Nunes C, et al. Effects of processing on physical characteristics of diets with distinct levels of carbohydrates and lipids: the outcomes on the growth of pacu (Piaractus mesopotamicus) [J]. Aquaculture Nutrition, 2010, 16(1): 91-99. doi: 10.1111/j.1365-2095.2008.00644.x
[16] Tan Q S, Xie S Q, Zhu X M, et al. Effect of dietary carbohydrate-to-lipid ratios on growth and feed utilization in Chinese longsnout catfish (Leiocassis longirostris Günther) [J]. Journal of Applied Ichthyology, 2007, 23(5): 605-610. doi: 10.1111/j.1439-0426.2007.00846.x
[17] Wang L N, Liu W B, Lu K L, et al. Effects of dietary carbohydrate/lipid ratios on non-specific immune responses, oxidative status and liver histology of juvenile yellow catfish Pelteobagrus fulvidraco [J]. Aquaculture, 2014(426): 41-48.
[18] Miao S, Nie Q, Miao H, et al. Effects of dietary carbohydrate-to-lipid ratio on the growth performance and feed utilization of juvenile turbot (Scophthalmus maximus) [J]. Journal of Ocean University of China, 2016, 15(4): 660-666. doi: 10.1007/s11802-016-2934-8
[19] Kabir K A, Verdegem M C J, Verreth J A J, et al. Effect of dietary carbohydrate to lipid ratio on performance of Nile tilapia and enhancement of natural food in pond aquaculture [J]. Aquaculture Reaserch, 2020, 51(5): 1942-1954. doi: 10.1111/are.14546
[20] Zhou Z, Ren Z, Zeng H, et al. Apparent digestibility of various feedstuffs for bluntnose black bream Megalobrama amblycephala Yih [J]. Aquaculture Nutrition, 2008, 14(2): 153-165. doi: 10.1111/j.1365-2095.2007.00515.x
[21] Yang Z F, Xu G C, Ge X P, et al. The effects of crowding stress on the growth, physiological response, and gene expression of the Nrf2-Keap1 signaling pathway in blunt snout bream (Megalobrama amblycephala) reared under in-pond raceway conditions [J]. Comparative Biochemistry and Physiology Part A: Molecular & Integrative Physiology, 2019(231): 19-29.
[22] 中华人民共和国农业农村部. 中国渔业统计年鉴 [M]. 北京: 中国农业出版社, 2019: 25. Ministry of Agriculture of the People’s Republic of China. Chinese Fishery Statistical Yearbook [M]. Beijing: Chinese Agricultural Press, 2019: 25.
[23] Zhou C, Liu B, Ge X, et al. Effect of dietary carbohydrate on the growth performance, immune response, hepatic antioxidant abilities and heat shock protein 70 expression of Wuchang bream, Megalobrama amblycephala [J]. Journal of Applied Ichthyology, 2013, 29(6): 1348-1356. doi: 10.1111/jai.12264
[24] Li X, Liu W, Jiang Y, et al. Effects of dietary protein and lipid levels in practical diets on growth performance and body composition of blunt snout bream (Megalobrama amblycephala) fingerlings [J]. Aquaculture, 2010, 303(1): 65-70.
[25] Ren M, Habte-Tsion H M, Xie J, et al. Effects of dietary carbohydrate source on growth performance, diet digestibility and liver glucose enzyme activity in blunt snout bream, Megalobrama amblycephala [J]. Aquaculture, 2015(438): 75-81. doi: 10.1016/j.aquaculture.2015.01.008
[26] Zhou C, Ge X, Liu B, et al. Effect of dietary carbohydrate level on growth performance, blood chemistry, hepatic enzyme activity, and growth hormone gene expression in Wuchang bream (Megalobrama amblycephala) [J]. The Israeli Journal of Aquaculture-Bamidgeh, 2013, 65(1): 1-8.
[27] Wang B K, Liu W B, Xu C, et al. Dietary carbohydrate levels and lipid sources modulate the growth performance, fatty acid profiles and intermediary metabolism of blunt snout bream Megalobrama amblycephala in an interactive pattern [J]. Aquaculture, 2017(481): 140-153. doi: 10.1016/j.aquaculture.2017.08.034
[28] Li X F, Wang Y, Liu W B, et al. Effects of dietary carbohydrate/lipid ratios on growth performance, body composition and glucose metabolism of fingerling blunt snout bream Megalobrama amblycephala [J]. Aquaculture Nutrition, 2013, 19(5): 701-708. doi: 10.1111/anu.12017
[29] AOAC. Official Methods of Analysis of the Association of Official Analytical Chemists [M]. 17th edn. Arlington: Association of Official Analytical Chemists, 2003: 1298.
[30] Folch J F. A simple method for the isolation and purification of total lipids from animal tissues [J]. Journal of Biological Chemistry, 1957, 22(226): 497-509.
[31] Du Z Y, Liu Y J, Tian L X, et al. Effect of dietary lipid level on growth, feed utilization and body composition by juvenile grass carp (Ctenopharyngodon idella) [J]. Aquaculture Nutrition, 2005, 11(2): 139-146. doi: 10.1111/j.1365-2095.2004.00333.x
[32] Hilton J W, Atkinson J L. Response of rainbow trout (Salmo gairdneri) to increased levels of available carbohydrate in practical trout diets [J]. British Journal of Nutrition, 1982, 47(3): 597-607. doi: 10.1079/BJN19820071
[33] Hemre G I, Sandnes K, Lie Ø, et al. Carbohydrate nutrition in Atlantic salmon, Salmo salar L.: growth and feed utilization [J]. Aquaculture Research, 1995, 26(3): 1 49-154. doi: 10.1111/j.1365-2109.1995.tb00896.x
[34] Tian L X, Liu Y J, Yang H J, et al. Effects of different dietary wheat starch levels on growth, feed efficiency and digestibility in grass carp (Ctenopharyngodon idella) [J]. Aquaculture International, 2012, 20(2): 283-293. doi: 10.1007/s10499-011-9456-6
[35] Vásquez-Torres W, Arias-Castellanos J A. Effect of dietary carbohydrates and lipids on growth in cachama (Piaractus brachypomus) [J]. Aquaculture Research, 2013, 44(11): 1768-1776.
[36] Li X F, Lu K L, Liu W B, et al. Effects of dietary lipid and carbohydrate and their interaction on growth performance and body composition of juvenile blunt snout bream, Megalobrama amblycephala [J]. The Israeli Journal of Aquaculture–Bamidgeh, 2014(66): 931-938.
[37] Brauge C, Medale F, Corraze G. Effect of dietary carbohydrate levels on growth, body composition and glycaemia in rainbow trout, Oncorhynchus mykiss, reared in seawater [J]. Aquaculture, 1994, 123(1–2): 109-120.
[38] Lanari D, Poli B M, Ballestrazzi R, et al. The effects of dietary fat and NFE levels on growing European sea bass (Dicentrarchus labrax L.) growth rate, body and fillet composition, carcass traits and nutrient retention efficiency [J]. Aquaculture, 1999, 179(1): 351-364.
[39] Shimeno S, Ming D C, Takeda M. Metabolic response to dietary carbohydrate to lipid ratios in Oreochromis niloticus [J]. Nippon Suisan Gakkaishi, 1993, 59(5): 827-833. doi: 10.2331/suisan.59.827
[40] Ali M, Jauncey K J A I. Optimal dietary carbohydrate to lipid ratio in African catfish Clarias gariepinus (Burchell 1822) [J]. Aquaculture International, 2004, 12(2): 169-180. doi: 10.1023/B:AQUI.0000032065.28059.5b
[41] Uyeda K, Repa J J. Carbohydrate response element binding protein, ChREBP, a transcription factor coupling hepatic glucose utilization and lipid synthesis [J]. Cell Metabolism, 2006, 4(2): 107-110. doi: 10.1016/j.cmet.2006.06.008
[42] Li H Y, Xu W J, Jin J Y, et al. Effects of dietary carbohydrate and lipid concentrations on growth performance, feed utilization, glucose, and lipid metabolism in two strains of gibel carp [J]. Frontiers in Veterinary Science, 2019(6): 165. doi: 10.3389/fvets.2019.00165
[43] 刘梅珍, 石文雷, 朱晨炜, 等. 饲料中脂肪的含量对团头鲂鱼种生长的影响 [J]. 水产学报, 1992, 16(4): 330-336. Liu M Z, Shi W L, Zhu C W, et al. The effect of dietary lipid level on the growth of blunt snout bream (Megalgerama amelycephala) fingerling [J]. Journal of Fisheries of China, 1992, 16(4): 330-336.
[44] McCue M D. Starvation physiology: Reviewing the different strategies animals use to survive a common challenge [J]. Comparative Biochemistry and Physiology Part A: Molecular & Integrative Physiology, 2010, 156(1): 1-18.
[45] Lu K L, Xu W N, Li J Y, et al. Alterations of liver histology and blood biochemistry in blunt snout bream Megalobrama amblycephala fed high-fat diets [J]. Fishereis Science, 2013, 79(4): 661-671. doi: 10.1007/s12562-013-0635-4
[46] Yan J, Liao K, Wang T, et al. Dietary lipid levels influence lipid deposition in the liver of large yellow croaker (Larimichthys crocea) by regulating lipoprotein receptors, fatty acid uptake and triacylglycerol synthesis and catabolism at the transcriptional level [J]. PLoS One, 2015, 10(6): e0129937. doi: 10.1371/journal.pone.0129937
[47] Du Z Y, Clouet P, Zheng W H, et al. Biochemical hepatic alterations and body lipid composition in the herbivorous grass carp (Ctenopharyngodon idella) fed high-fat diets [J]. British Journal of Nutrition, 2006, 95(5): 905-915. doi: 10.1079/BJN20061733
[48] Kikuchi K, Furuta T, Iwata N, et al. Effect of dietary lipid levels on the growth, feed utilization, body composition and blood characteristics of tiger puffer Takifugu rubripes [J]. Aquaculture, 2009, 298(1): 111-117.
-
期刊类型引用(1)
1. 刘伟,蒋明,吴凡,田娟,陆星,喻丽娟,文华. 饲料能量密度和投喂水平对吉富罗非鱼生长和健康的影响. 水生生物学报. 2022(01): 58-68 .  本站查看
本站查看
其他类型引用(1)



 下载:
下载: